mRNA Vaccine Risks
48.2K Healthcare Employee Study—The More Doses with COVID-19 mRNA Vaccine, the Higher the Infection Risk
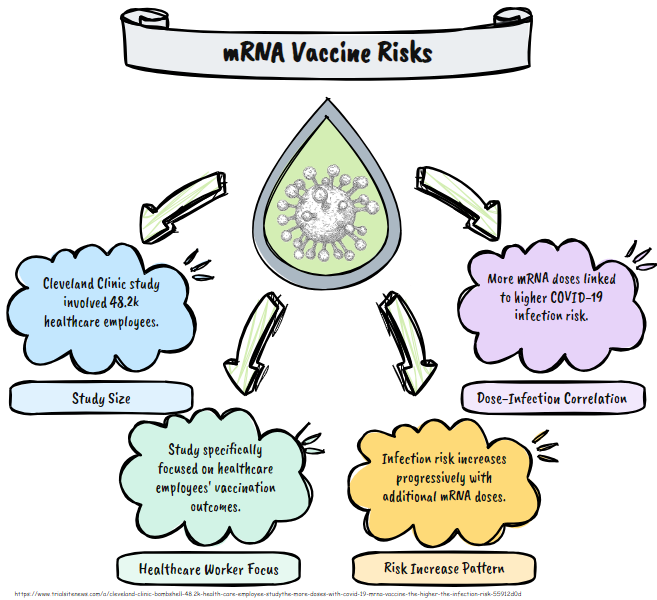
A study conducted by Cleveland Clinic researchers involving 48,211 employees to assess the effectiveness of the 2023-2024 formulation of the COVID-19 mRNA vaccine against the JN.1 variant of SARS-CoV-2. The study, which ran for 17 weeks, found that 5.1% of participants (2,462 employees) contracted COVID-19. It reported that the vaccine provided modest protection before the JN.1 lineage became dominant, with a hazard ratio of 0.58 (indicating a significantly lower risk), but this protection diminished afterward, with a hazard ratio of 0.81 (not statistically significant).
A key finding highlighted in the article is an association between the number of prior mRNA vaccine doses and an increased risk of COVID-19 infection. Specifically, the risk was incrementally higher with more doses: compared to those with 0 or 1 doses, the hazard ratio was 1.46 for 2 doses, 1.75 for 3 doses, and 2.0 for 4 or more doses. The article suggests this raises questions about the long-term impact of multiple vaccine doses, though it notes the study's authors did not attribute a definitive cause, such as potential immune system effects. The vaccine's overall effectiveness against JN.1 was estimated at 23%, leading the article to characterize the findings as a "bombshell" that challenges assumptions about repeated boosters.
"Are We Training Our Immune System to Surrender? The Shocking Rise of IgG4 After Pfizer’s mRNA Vaccination," explores emerging research suggesting that repeated doses of Pfizer’s mRNA COVID-19 vaccine may lead to an unexpected immunological shift. Published on February 20, 2025, it focuses on a study titled "IgG4 Antibodies Induced by mRNA Vaccines Generate Immune Tolerance to SARS-CoV-2’s Spike Protein by Suppressing the Immune System," which posits that the rise in IgG4 antibodies following multiple mRNA vaccinations could indicate immune tolerance rather than enhanced protection. The article highlights that IgG4, a subclass of immunoglobulin G typically associated with anti-inflammatory responses (e.g., in allergen immunotherapy or chronic infections), increased significantly after the second and third Pfizer doses. Unlike IgG1 and IgG3, which dominate early immune responses and are pro-inflammatory, IgG4 does not effectively activate complement or engage immune effector cells, potentially weakening antiviral defenses. The study suggests that this shift might allow SARS-CoV-2 to replicate more easily by suppressing natural immune responses, raising concerns about increased susceptibility to infections, autoimmune diseases, and even cancer progression in vulnerable individuals. It contrasts this with adenoviral vector vaccines (e.g., AstraZeneca), which did not show similar IgG4 elevations, suggesting a unique effect of mRNA platforms. The article frames these findings as a potential challenge to the narrative of booster efficacy, urging further investigation into long-term implications.
Key Findings and Mechanisms The article delves into the observation that IgG4 levels rose in about half of Pfizer vaccine recipients seven months after their second dose, with a near-universal increase following a third booster. This shift is unusual because IgG4 is not typically prominent in responses to viral infections or traditional vaccines like tetanus or RSV, where repeated antigen exposure does not trigger such a class switch. The authors of the referenced study propose that excessive antigen concentration from mRNA vaccines—delivered via lipid nanoparticles encoding the SARS-CoV-2 spike protein—combined with repeated dosing, drives this immunological change. Unlike adenoviral vaccines, which showed minimal IgG4 induction, mRNA vaccines appear to uniquely promote this tolerance mechanism. IgG4’s functional properties amplify the article’s concerns. Its inability to bind complement or Fc receptors on immune cells (like macrophages or NK cells) means it may act as a "blocking antibody," potentially outcompeting more effective IgG subclasses. This could dampen the immune system’s ability to clear SARS-CoV-2, leading to what the article dramatically calls "training our immune system to surrender." The study also notes that prior SARS-CoV-2 infection seems to mitigate this IgG4 rise, suggesting that natural immunity might "prime" the immune system differently than vaccination alone—an observation that aligns with debates about hybrid immunity.
Broader Implications The article expands on the study’s speculation about downstream health risks. It cites suggestions that elevated IgG4 could contribute to autoimmune conditions (e.g., myocarditis) by disrupting immune regulation or promote cancer growth by suppressing antitumor immunity—a hypothesis linked to IgG4’s role in chronic antigen exposure scenarios. These claims, while provocative, are presented as emerging theories requiring further research, reflecting the article’s tone of alarm tempered by calls for scientific scrutiny. The piece also critiques the mRNA vaccination strategy itself. It notes that Pfizer and Moderna boosters maintain high antigen doses, contrary to traditional vaccinology principles that favor lower doses for boosting to avoid immune exhaustion. This, combined with the persistence of mRNA-encoded spike protein in the body (detected in lymph nodes months later), might overstimulate the immune system, pushing it toward tolerance rather than robust defense.
Comparison with Other Vaccines A notable contrast is drawn with adenoviral vector vaccines (e.g., AstraZeneca) and protein-based vaccines (e.g., Novavax), which do not elicit significant IgG4 responses. This suggests that the mRNA platform’s mechanism—direct cellular production of spike protein—may uniquely influence antibody class switching. The article implies that this difference could inform future vaccine design, questioning the one-size-fits-all approach to COVID-19 boosters.
Dig In
Are we training our immune system to surrender? Cleveland Clinic Bombshell Yale Study
